Until InterStim was available, patients with urgency, frequency, urge incontinence or non-obstructive urinary retention who did not respond to traditional and/or medical therapy was left with little recourse.
About InterStim
Medtronic InterStim® System for Urinary Control is indicated for the treatment of urinary retention and the symptoms of overactive bladder, including urinary urge incontinence and significant symptoms of urgency-frequency alone or in combination, in patients who have failed or could not tolerate more conservative treatments.
InterStim Therapy uses a small device to send mild electrical pulses to a nerve located in the lower back (just above the tailbone).
This nerve, called the sacral nerve, influences the bladder and surrounding muscles that manage urinary function. The electrical stimulation may eliminate or reduce certain bladder control symptoms in some people. The system is surgically placed under the skin.
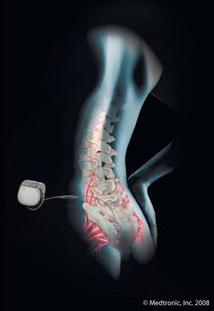
A significant benefit of InterStim Therapy is the test stimulation. Before a decision to sugically place the stimulator under the skin, the patient has the opportunity to test if may work. This outpatient procedure assesses the effect of InterStim Therapy for each patient prior to consideration of a surgical implant procedure.
The test stimulation:
- demonstrates the effect of sacral nerve stimulation on patient symptoms over a 3-5 day trial period
- allows the patient to experience the sensation of stimulation during various everyday activities
- helps the physician make an informed choice about InterStim Therapy as a long-term therapy option
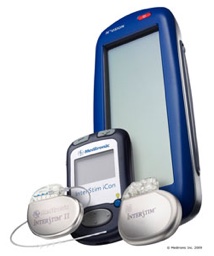 Within three to five days, the patient can determine if InterStim Therapy is a viable treatment option. During the test stimulation, patients are asked to keep a voiding diary to record voiding behavior and symptoms with the stimulation. The voiding diary is then compared with baseline and post-test stimulation diaries to determine the effect of sacral nerve stimulation on symptoms.
Within three to five days, the patient can determine if InterStim Therapy is a viable treatment option. During the test stimulation, patients are asked to keep a voiding diary to record voiding behavior and symptoms with the stimulation. The voiding diary is then compared with baseline and post-test stimulation diaries to determine the effect of sacral nerve stimulation on symptoms.
The test stimulation allows the patient to evaluate the therapy as an option before the commitment of an implant. It also provides patients with realistic expectations about the results of InterStim Therapy.
If the test stimulation is successful, the patient may go on to the implantation of the InterStim System.
